Lokelma, the brand name of Sodium Zirconium Cyclosilicate, is developed and marketed by AstraZeneca pharmaceutical company. It got FDA approval in 2018 for the treatment of hyperkalemia.
Lokelma (Sodium Zirconium Cyclosilicate) is a non-absorbable potassium binder. As the name suggests, it contains sodium which along with hydrogen ions is exchanged for potassium ions in the gut.
Thus, it inhibits the potassium ions from getting absorbed and going into the systemic circulation. Unlike Kayexalate powder, it is highly specific and has little effect on the absorption of other ions like calcium and magnesium.
| You may also like to read: |
Lokelma (Sodium Zirconium Cyclosilicate) Uses (Indications):
Lokelma (Sodium Zirconium Cyclosilicate) is indicated for the treatment of hyperkalemia. It inhibits the potassium from getting absorbed into the systemic circulation.
Therefore, it is not indicated in patients who need acute or emergency treatment of hyperkalemia.
The plasma potassium starts to decrease within one hour of administering Lokelma. However, for optimal results, it may take 48 to 72 hours to normalize serum potassium levels.
In one clinical trial, the plasma potassium normalized in 82% of the participants within 24 hours and 99% of the patients after 72 hours [Ref].
After 72 hours, patients were initiated on long-term maintenance treatment of hyperkalemia.
| You may also like to read: |
Lokelma (Sodium Zirconium Cyclosilicate) Dosage form and Dosing:
Lokelma (Sodium Zirconium Cyclosilicate) is available as an odorless powder in foiled packets. Each packet contains either 5 gms or 10 gms of Sodium Zirconium Cyclosilicate.
Initially, it is administered in a higher dose until the potassium in the plasma drops to normal after which the maintenance dose can be given either daily or on alternate days.
Initial correction dose for Hyperkalemia:
- It is given in a dose of 10 gms powder (as an oral suspension in plain water) three times a day for the first 48 hours (or at most 72 hours). It is usually given before a meal to effectively bind all the dietary potassium.
Maintenance dose for Hyperkalemia:
- The usual maintenance dose of hyperkalemia is 10 gms once daily. The dose is adjusted at weekly intervals based on the plasma potassium levels.
- The minimum recommended maintenance dose can be as low as 5 gms every other day to as high as 15 gm once daily.
- Treatment may be discontinued if the potassium levels fall below the normal ranges.
Dose in patients who are on hemodialysis:
- For patients on hemodialysis, it is best to administer Sodium Zirconium Cyclosilicate on non-dialysis days.
- Treatment may be initiated if the serum potassium exceeds 6.5 mEq/L on non-dialysis days.
- It is given initially in a dose of 10 gm once daily on non-dialysis days.
- Dose titration is done at weekly intervals. The usual potassium dose may range from 5 gm to 15 gm once a day on non-dialysis days.
- If the patient develops significant hypokalemia, treatment should be discontinued.
| You may also like to read: |
How to administer Lokelma (Sodium Zirconium Cyclosilicate)?
Sodium Zirconium Cyclosilicate is available in the form of powder. It is taken orally by first making a suspension.
The powder is mixed with plain water in a glass. A minimum of 45 ml or three tablespoons of plain water is mixed with the desired amount of Lokelma powder.
The suspension is stirred to mix the contents properly and taken orally immediately. If any of the content remains in the glass, a little water is added again, and drink it immediately until all the medicine is ingested.
It is best to take the drug before breakfast if you take it only once a day, or before each meal if you take it three times a day.
If you are on concomitant medications, taking Lokelma at least two hours before or two hours after the drug is best.
Simultaneous administration of Lokelma and other medications might impair the absorption of the concomitant medicine.
| You may also like to read: |
Lokelma (Sodium Zirconium Cyclosilicate) Dose in Pregnancy and Breastfeeding:
Since Lokelma does not enter systemic circulation, it is safe to use in pregnant and breastfeeding women.
No dosage adjustments have been recommended in these patients.
Lokelma (Sodium Zirconium Cyclosilicate) Dose in Kidney and Liver diseases:
Since it does not enter the systemic circulation, dose adjustment is not necessary. However, in patients who are undergoing hemodialysis, it is recommended only on predialysis days.
The incidence of hypokalemia is high in these patients and frequent monitoring of serum potassium levels is recommended followed by dose adjustment according to the potassium levels.
In patients with clinically significant hypokalemia, the treatment may be discontinued for some time followed by reinitiation of the dose at a lower dose.
| You may also like to read: |
Lokelma (Sodium Zirconium Cyclosilicate) Side effects and Contraindications:
The manufacturer has not recommended any contraindications to Sodium Zirconium Cyclosilicate.
However, in certain conditions, especially related to the gastrointestinal tract, Lokelma should be used with caution. These include:
Individuals with GI motility disorders:
One thing is clear, if you don’t poop after taking Lokelma, you are not going to get rid of potassium. So, in patients with constipation or intestinal obstruction, the drug may become ineffective.
Secondly, it has not been studied in clinical trials in patients with GI motility disorders like constipation, intestinal obstruction, stool impaction, or post-operative motility disorders of the gut.
It may worsen the underlying GI disorder and hence is not recommended in such conditions.
Edema:
As the name suggests, Sodium Zirconium Cyclosilicate, Lokelma contains sodium. Each 5 gm of Lokelma powder contains about 400 mg of Sodium [Ref].
The usual daily recommendations or dietary allowance for sodium intake in kidney and heart diseases are:
- 750 to 2000 mg per day for patients with chronic kidney disease
- 1500 mg for patients with heart diseases
- A maximum of 2300 mg for people without kidney or heart disease
If you take 10 gm three times a day, your sodium intake from Lokelma will equal 2400 mg. This even exceeds the daily recommended sodium intake of 2300 mg for people without any medical condition.
In clinical trials, patients with renal failure who took Lokelma on non-dialysis days in doses of 5 gm or 10 gm did not observe any weight gain compared to those who were not taking it.
Since weight gain is a sign of fluid overload and excess sodium intake, if taken in low doses of up to 10 gm on non-dialysis days, it may not cause fluid retention.
However, the chances of a fluid overload state are very high while you are taking Lokelma, it is therefore recommended to adjust your dietary sodium intake. This is especially true if you are on the initial high dose for the correction of hyperkalemia.
Patients with impaired left ventricular function are at an increased risk of developing heart failure. Sodium restriction in the diet should be strictly advised while on Lokelma.
Hypokalemia:
Since Sodium Zirconium Cyclosilicate is used to lower potassium in the blood, the chances of hypokalemia are there.
Patients who are on hemodialysis and those who develop other medical conditions like gastroenteritis, diarrhea, vomiting, or are on potassium-losing diuretics (Furosemide) may develop hypokalemia.
Potassium levels, especially in these patients should be strictly monitored and the dose of Lokelma should be adjusted accordingly.
| You may also like to read: |
Side effects of Lokelma (Sodium Zirconium Cyclosilicate):
Edema:
Lokelma is a non-absorbable drug that has minimal side effects. However, the compound contains a lot of sodium which can get absorbed into the system and cause fluid retention, worsen hypertension, and may precipitate heart failure.
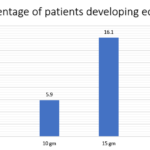
In clinical trials, edema was found in a greater percentage of patients who took the drug in higher doses.
Compared to the placebo, edema was significant in patients who took Lokelma in a dose of 10 gm and 15 gm per day.
Lokelma Dose | Percentage of patients developing edema |
| 5 gm | 4.4 |
| 10 gm | 5.9 |
| 15 gm | 16.1 |
| Placebo | 2.4 |
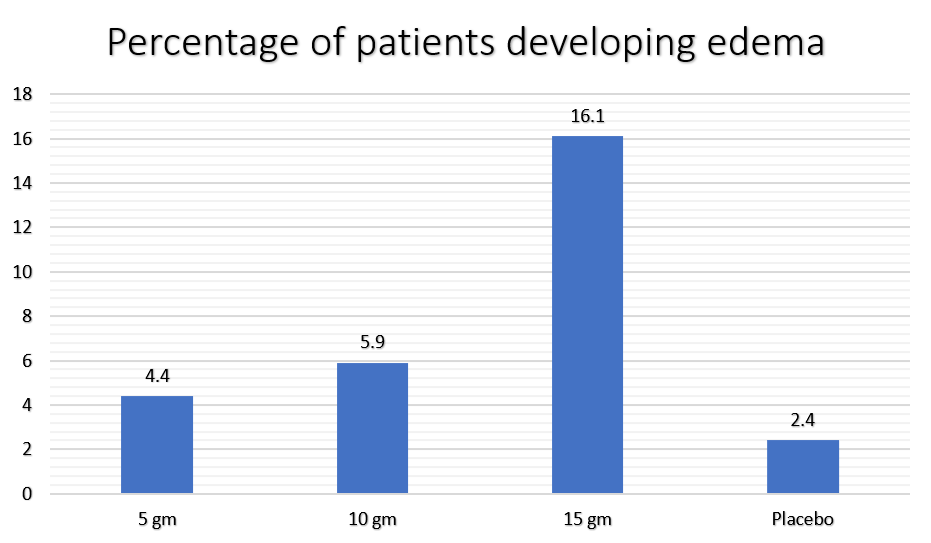
The prevalence of edema is 8% to 11% in patients who take it in a dose of less than 15 gm per day for prolonged periods.
Hypokalemia:
Hypokalemia can occur since the drug inhibits potassium absorption. However, with proper dose adjustment, it can be avoided.
Hypokalemia can occur in patients who develop diarrhea, vomiting, or are on medications that may lower the plasma potassium levels.
Hypokalemia is defined as a serum potassium level that is less than 3.5 mEq/L. The incidence of hypokalemia is estimated to be around 4.1% of the patients on Lokelma treatment.
In patients with chronic kidney disease who are on maintenance hemodialysis, the incidence of pre-dialysis hypokalemia was:
Patients on Lokelma | Placebo | |
| K < 3.5 mEq/L | 5% | 5% |
| K < 3 mEq/L | 3% | 1% |

| You may also like to read: |
Mechanism of action of Lokelma (Sodium Zirconium Cyclosilicate MOA):
Sodium Zirconium Cyclosilicate is a non-absorbable drug that acts by inhibiting potassium absorption from the gut.
It exchanges hydrogen and sodium ions for Potassium. It preferentially binds to potassium even in the presence of other cations such as calcium and magnesium ions.
As it captures potassium, more potassium gets excreted with it in the feces. This lowers the plasma potassium levels.
The potassium-lowering effects of Lokelma depend on the dose of the drug. The higher the dose, the more potassium will it bind and ultimately excrete via feces.
The onset of action:
The potassium-lowering effect of Lokelma can be observed within one hour after the drug is administered. When given in three doses per day for 48 hours, the potassium-lowering effect continues.
It has also been seen that the greater the serum potassium or the higher the dose of Lokelma is, the more potassium will be excreted. This will lead to a greater reduction in serum potassium levels.
It has also been observed that Lokelma causes an insignificant increase in plasma bicarbonate levels in a dose-dependent manner.
The increase in plasma bicarbonate levels is as mentioned in the table below:
Lokelma Dose | The rise in Bicarbonate levels in the plasma |
| 5 gm | 1.1 mmol/L |
| 10 gm | 2.3 mmol/L |
| 15 gm | 2.6 mmol/L |
| Placebo | 0.6 mmol/L |
Absorption:
It does not get absorbed and works locally.
Metabolism:
It is an insoluble and inorganic molecule that is neither absorbed nor metabolized by proteolytic enzymes.
| You may also like to read: |
Lokelma Drug Interactions:
Lokelma alters the PH of the gastric and intestinal lumen. It increases the luminal PH.
It has been observed that Lokelma may alter the absorption of medications that require an optimal PH for their absorption.
Therefore, for medications that are absorbed into the systemic circulation from the GI tract in a PH-dependent manner, their absorption might get affected.
The manufacturer tested the absorption of 36 drugs from the GI tract while the patients received Lokelma simultaneously.
The absorption of 16 drugs was not affected while 20 showed in vitro interactions. Out of the 20 drugs that showed in vitro interactions, 9 were then tested in healthy volunteers.
The results of the 9 tested drugs in healthy volunteers have been tabulated below [Ref]:
Absorption not affected by Lokelma | Absorption Decreased | Absorption Increased |
|
|
|
Lokelma (Sodium Zirconium Cyclosilicate) Cost:
The per unit or per sachet cost of 5 gms is around $ 25 USD.
The price per 10 gm sachet is roughly the same.