Wegovy Injections are similar to Ozempic injections except that the dose of Semaglutide administered is higher when Wegovy is used.
In other words, Wegovy is a high-dose injectable Semaglutide, like Ozempic, that is administered weekly.
Unlike all other GLP-1 analogs, Semaglutide is 94% identical to the natural endogenous GLP-1. It has the smallest size and can also cross the blood-brain barrier.
Because of its low molecular weight, its weight loss effects are directly a result of its effects on the satiety center in the brain, apart from its direct effects on the gastrointestinal tract.
Wegovy Uses and Indications [Ref]:
Wegovy is FDA-approved for the treatment of obesity and weight management. It should be used as an adjunct to a low-calorie diet and exercise.
Wegovy is used for the treatment of weight loss in the following individuals:
- Those whose BMI is equal to or exceeds 30 kg/m². These patients are generally categorized as obese individuals.
- Those people with a BMI of 27 kg/m² or more in the presence of at least one obesity-related complication, such as Diabetes, Hypertension, and Dyslipidemia. These patients are generally classified as “overweight” individuals.
Wegovy is not to be used in the following patients:
- It should be avoided in patients with a history of pancreatitis.
- It is not recommended to be used in combination with other GLP-1 analogs like Liraglutide (Saxenda), Dulaglutide (Trulicity), or DPP-IV inhibitors like Januvia and Galvus.
- Wegovy has not been studied in patients who are using other weight-loss drugs. These may include:
- Prescription weight-loss drugs,
- Over-the-counter weight loss drugs, and
- Herbal products for weight loss
Wegovy Starting Dose, Usual Dose, and Dose Escalation:
Wegovy use is associated with significant gastrointestinal side effects. To minimize the GI side effects, it is initially administered in low doses and then gradually titrated up to the maximum tolerated dose.
Wegovy Starting Dose:
- 0.25 mg subcutaneous injection, administered every week for four weeks,
Wegovy Dose Escalation dose:
- After the initial four weeks, the dose is increased at four-weekly intervals.
- From week 5 to 8, it is administered in a dose of 0.5 mg sub Q weekly,
- From week 9 to 12, a dose of 1 mg is administered weekly,
- From week 13 to16, a dose of 1.7 mg is administered weekly, and
- From week 17 onwards, the maximum dose of 2.4 mg is administered weekly.
The following table summarizes the dose of Wegovy:
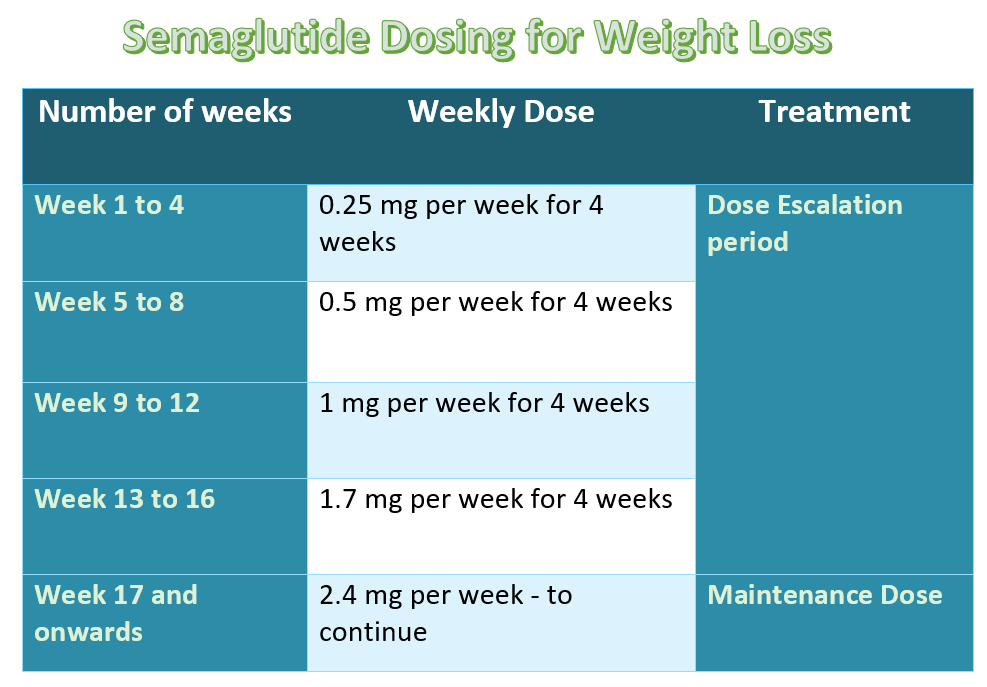
The maximum dose of Wegovy is 2.4 mg subcutaneously once weekly. However, if a person can not tolerate the maximum (usual) dose of Wegovy, a lower dose of 1.7 mg may be continued for a while (for an additional 4 to 8 weeks) and the dose should then be increased.
Similarly, during the dose-escalation period, if a person is not tolerating the higher dose, he/ she should be continued for four additional weeks on the lower dose, after which the dose should be titrated.
All patients with diabetes should have their blood sugars monitored during Wegovy treatment. If the usual dose can not be tolerated at all, treatment should be discontinued.
If you want to change your Wegovy dosing schedule …
If you want to change your weekly schedule of Wegovy injections, you can switch to another day at your ease, however, the time between two doses should be kept to a minimum of 48 hours (two days).
If you miss your Wegovy dose, …
If you miss your dose of Wegovy, you can administer it as soon as possible and within five days of the last scheduled dose.
However, if more than five days have passed, skip that dose and administer the dose on the next scheduled day.
For example, you inject Wegovy every Sunday. Now, you missed this Sunday’s dose. You can administer it as soon as you remember, before Friday.
If you remember on Friday or Saturday that you missed your dose, you should skip it and administer your next dose on your scheduled day (Sunday).
Your weekly schedule will resume thereafter.
How do you administer Wegovy Injections?
Wegovy injections are administered once a week into the skin of:
- Belly (3 to 5 cm away from the umbilicus)
- Thighs, and
- Upper arms
The injection site should be repeated every time. It is recommended to inspect the contents of the injection. If the solution is cloudy, opaque, or discolored, it should not be used.
Wegovy should not be injected into the skin where insulin or other medications have been injected. It should not be mixed with other injections.
How is Wegovy Injection provided?
Wegovy injection is provided as single pre-filled syringes in doses of:
- 0.25 mg per 0.5 ml
- 0.5 mg per 0.5 ml
- 1 mg per 0.5 ml
- 1.7 mg per 0.75 ml, and
- 2.4 mg per 0.75 ml
Wegovy Contraindications and Warnings:
Wegovy is contraindicated if a person is allergic to Semaglutide or any component of the formulation.
Individuals with a history of serious allergic reactions, such as angioedema and anaphylaxis, should not use Semaglutide.
Wegovy is also contraindicated in patients with a personal or family history of MEN-2 syndrome (multiple endocrine neoplasia type 2).
Medullary Thyroid Cancer:
The use of GLP-1 analogs has been associated with an increased incidence of thyroid cancers. The risk of Medullary thyroid cancer with or without the presence of MEN-2 syndrome is high in patients on GLP-1 analogs.
The association has been observed in animal studies and post-marketing reports of patients receiving other GLP-1 analogs like Liraglutide.
Routine monitoring for the development of medullary thyroid cancer is not recommended, however, all patients who observe neck swelling or pain or have difficulty swallowing should be asked to report it.
Serum calcitonin levels and fine-needle aspiration may be advised if a person develops neck swelling or dysphagia while on Wegovy treatment.
Serum calcitonin level exceeding 50 ng/L is usually considered risky. These patients must be investigated for medullary thyroid cancer.
Acute Pancreatitis:
Acute Pancreatitis can occur in any individual who is using a GLP-1 analog. Patients should be asked to stop the treatment immediately if they develop acute pancreatitis.
Patients with acute pancreatitis can develop a sudden onset of severe epigastric pain that radiates to the back. The pain may be associated with nausea and vomiting as well.
All patients must be given full supportive treatment to reduce the complications associated with pancreatitis. In addition, apart from stopping Wegovy, all medications and fatty meals that may trigger pancreatitis should also be discontinued.
Patients with a past history of pancreatitis should not be treated with Semaglutide or another GLP-1 analog.
Acute Gallbladder Disease:
Wegovy use was associated with an increased risk of gallbladder disease in clinical trials. Patients on Wegovy are at a high risk of developing gallstones and inflammation of the gallbladder (cholecystitis).
The percentage of patients developing gallbladder disease is given in the table below:
| Gall Stones | Cholecystitis | |
| Wegovy | 1.6% | 0.6% |
| Placebo | 0.7% | 0.2% |
Although rapid weight loss is itself associated with gallstones, the risk was still high after accounting for the degree of weight loss.
Hypoglycemia:
Wegovy can cause hypoglycemia. Hypoglycemia is especially concerning if the person is also on concomitant insulin or other insulin-secreting drugs like sulfonylureas.
In clinical trials of overweight individuals (BMI of 27 or more), hypoglycemia was documented in 6.2% of the studied patients using Wegovy and 2.5% of those using a placebo. Hypoglycemia in clinical trials was defined as a blood glucose of 54 mg/dl or less.
Semaglutide in doses of 0.5 mg and 1 mg has been associated with more frequent hypoglycemia in combination with insulin and sulfonylureas. Hence, with Wegovy, the risk of hypoglycemia is much greater.
All patients who are initiated on Semaglutide treatment should be warned about the symptoms of hypoglycemia. Diabetic patients on insulin or other anti-diabetic medications should be especially warned.
Diabetic Retinopathy:
Semaglutide use has been associated with an increased incidence of diabetic retinopathy. In clinical trials, the incidence of diabetic retinopathy was 4% in patients using Wegovy compared to 2.7% in the placebo group.
Retinopathy has also been observed when Semaglutide is administered at a low dose of 0.5 mg and 1 mg weekly. In clinical trials, 3% of the patients using Ozempic and 1.8% in the placebo group developed retinopathy.
Retinopathy has been observed with insulin and other diabetes medicines as well. It is thought to occur as a result of the rapid improvement in blood glucose.
Furthermore, the risk of worsening diabetic retinopathy is greater in patients with a background disease. With Ozempic, it was 8.2% compared to 5.2% in the placebo group.
In patients without a history of diabetic retinopathy, the risk of diabetic retinopathy was 0.7% in the Ozempic-treated patients vs 0.4% in the placebo-treated patients.
It is recommended to monitor patients for diabetic retinopathy if they develop blurring or new-onset eye-related symptoms.
Avoid Pen-sharing:
Ozempic pen should not be shared by two individuals, even if the needle is changed. There is a risk of infection transmission from one person to another.
Acute renal failure:
Semaglutide use has been associated with acute renal failure. Renal failure is commonly seen in those patients who had abnormal renal function at baseline.
In addition, renal failure occurs commonly in patients with more severe gastrointestinal side effects such as nausea, vomiting, and dehydration.
However, the new onset of kidney dysfunction has also been reported
Individuals who initiate treatment using Semaglutide should be monitored for worsening renal function. Treatment should be discontinued if renal failure is observed.
Hypersensitivity reactions:
Semaglutide, like other GLP-1 analogs, may cause serious allergic reactions. Patients have been reported to develop angioedema and anaphylaxis.
Those with a previous history of anaphylaxis or serious allergic reactions to Ozempic or other GLP-1 should not be advised to use the drug.
In case of any reaction, supportive care should be promptly initiated. Patients may be given intravenous fluids, antihistamines, corticosteroids, and/or epinephrine.
Increased heart rate:
A racing heart rate has been reported in patients using Semaglutide. There is, on average 1 to 4 beat increase in patients using Semaglutide compared to placebo.
An increase in heart rate of 10 to 19 beats per minute was observed in 41% of the Wegovy-treated group vs 34% in the placebo-treated individuals.
An increase in the heart rate of more than 20 beats per minute was observed in 26% of the individuals treated with Wegovy compared to 16% of those treated with a placebo.
Patients may be asked to report if they have persistent palpitations and racing hearts.
Wegovy and suicidal risk:
Suicidal thoughts and ideation have been reported by obese individuals who have been treated with other weight loss medications or undergone any weight loss intervention.
Wegovy should not be initiated in people with suicidal risk and ideation. In addition, all patients should be monitored for the development of new-onset abnormal thoughts to inflict self-harm.
Wegovy Side Effects:
The most common side effects of Semaglutide injection are related to the gastrointestinal tract. These included nausea, vomiting, diarrhea, constipation, and abdominal pain.
In clinical trials, 73% of the patients treated with Weogvy vs 47% of those treated with a placebo medicine reported gastrointestinal side effects.
Some of the GI side effects were so severe that they led to treatment discontinuation. 4.3% of the patients treated with Wegovy vs 0.7% of those treated with a placebo medicine discontinued the treatment due to gastrointestinal side effects.
The relative percentage of these side effects is summarized in the table below:
| Side effects | Wegovy | Placebo |
| Nausea | 44% | 16% |
| Diarrhea | 30% | 16% |
| Vomiting | 24% | 6% |
| Constipation | 24% | 11% |
| Abdominal Pain | 20% | 10% |
| Headache | 14% | 10% |
| Fatigue | 11% | 5% |
| Dyspepsia | 9% | 3% |
| Dizziness | 8% | 4% |
| Abdominal Distension | 7% | 5% |
| Eructation | 7% | <1% |
| Hypoglycemia in T2DM | 6% | 2% |
| Flatulence | 6% | 4% |
| Gastroenteritis | 6% | 4% |
| Gastroesophageal Reflux Disease | 5% | 3% |
| Gastritis | 4% | 1% |
| Viral Gastroenteritis | 4% | 3% |
| Hair loss | 3% | 1% |
Other side effects, apart from those mentioned above and in the contraindications and warning sections, include:
Hypotension and Syncope:
Hypotension and syncope were reported in 1.3% of the patients treated with Wegovy compared to 0.4% of those treated with placebo medicine.
Similarly, symptoms of syncope were observed in 0.4% of the patients treated with Semaglutide vs 0.2% of those treated with placebo medicine.
Appendicitis:
Appendicitis, including perforated appendiciti,s was diagnosed in 10 (0.5%) of the patients receiving Wegovy vs 2 (0.2%) of the patients receiving placebo.
Injection-site reactions:
Injection site reactions were observed in 1.4% vs 1% in the Semaglutide and placebo groups, respectively.
Injection site reactions may vary from mild to severe. These may include itching, pruritus, erythema, inflammation, and induration.
Laboratory abnormalities:
Patients in the treatment group had raised serum amylase and lipase levels.
Serum amylase levels increased by 16% while serum lipase levels increased in 39% of the patients treated with Semaglutide compared to none in the placebo groups.
Fatigue, Altered taste, and dizziness:
- Fatigue is commonly reported in post-marketing studies; however, in clinical trials, the risk was estimated to be around 0.4%
- Similarly, altered taste and dizziness were also reported in a similar number of patients.
Wegovy Drug Interactions:
Semaglutide does not have any effects on drug transporter proteins. It does not have any effect on either CYP enzyme induction or enzyme inhibition.
However, Semaglutide causes delayed gastric emptying that may impair the absorption of certain drugs, including:
- Levothyroxine
- Warfarin
- Metformin
- Digoxin
- Atorvastatin
- Ethinylestradiol, and
- Levonorgestrel
The manufacturer, however, does not recommend any dose adjustments when combining these drugs with Semaglutide.
In addition, Wegovy, when administered with oral anti-diabetic drugs or insulin, may increase the risk of hypoglycemia. Diabetic patients may need adjustments in their diabetes medications.
Wegovy Use in Pregnancy and Breastfeeding:
Semaglutide has not been studied in human pregnancies. However, animal studies have resulted in structural defects, alterations in growth, and fetal deaths.
The risks of fetal malformations are also great in pregnant diabetic females who have uncontrolled diabetes.
Pregnant females who have been accidentally exposed to the drug during their pregnancy are encouraged to report to Novo Nordisk at 1-800-727-6500.
Use during breastfeeding:
Semaglutide use in lactating mothers has not been studied. In animals, the drug was found in the milk of lactating animals, however, it has not been studied in humans.
The manufacturer recommends weighing the benefits and risks of Semaglutide and breastfeeding before treatment initiation or continuation.
Males and females of reproductive potential:
Semaglutide should be discontinued at least two months before a planned pregnancy because of its long half-life.
Wegovy use in children and older individuals:
Semaglutide has not been studied in individuals younger than 18 years of age. The manufacturer does not recommend its use in individuals less than 18 years of age.
Older individuals (75 years of age or more) may be sensitive to the drug. The incidence of side effects, including hypoglycemia (especially when concomitant insulin or sulfonylureas are being used) may be greater in the older population.
In clinical trials, there was no difference in the safety or efficacy of Semaglutide in younger and older individuals, however, it should be used with caution in older people because of the increased sensitivity of the drug.
Wegovy use in patients with kidney diseases:
Semaglutide has been studied in patients with mild, moderate, severe, and ESRD (end-stage renal disease). Kidney disease does not affect the pharmacokinetics of Semaglutide.
Hence, dose adjustment in patients with kidney disease has not been recommended by the manufacturer.
Wegovy use in patients with liver diseases:
Semaglutide has been studied in varying degrees of liver disease. Liver disease does not affect the pharmacokinetics of Semaglutide.
Hence, dose adjustment has not been recommended by the manufacturer in patients with liver disease.
What to do if you overdose on Wegovy?
Overdose can happen. Patients can manifest severe gastrointestinal side effects such as:
- Nausea
- Vomiting
- Abdominal Pain
- Diarrhea, or Constipation
- Altered taste.
- Abdominal fullness.
The risk of severe and prolonged hypoglycemia is also there. Patients may need supportive treatment and prolonged monitoring because of the long half-life of the drug.
Other serious side effects of Ozempic overdose may include pancreatitis, renal failure, and anaphylaxis.
In any case, it is better to consult your healthcare provider. Treatment is usually supportive by adhering to a healthy diet, keeping yourself well-hydrated, and taking antacids.
Wegovy MOA (Mechanism of action) and Pharmacology:
Semaglutide is a GLP-1 agonist. It binds to the endogenous GLP-1 receptors that are present in various parts of the body and activates them. Semaglutide is 94% identical to the natural GLP-1.
Because of its low molecular weight, it can cross the blood-brain barrier and bind to the GLP-1 receptors in the brain.
It primarily suppresses the satiety centers in the brain by its direct effects. However, it also causes distension in the stomach.
Stretch receptors in the stomach, via their negative feedback mechanisms, can suppress satiety centers in the brain.
Semaglutide in low doses, as in Ozempic, is used to treat patients with diabetes.
Semaglutide enhances the secretion of insulin and lowers glucagon levels. These effects of Semaglutide are greatest when the blood glucose is high. Thus, Semaglutide lowers blood glucose in a glucose-dependent manner.
Semaglutide also inhibits or delays gastric emptying. This has a slight post-prandial glucose-lowering effect. However, this effect is primarily responsible for the weight loss effects.
Semaglutide does not have any effect on the QT interval.
Absorption:
- Semaglutide reaches its maximum plasma concentrations after 1 to 3 days of administration.
Steady-state concentration:
- Semaglutide reaches its steady-state concentration in about 4 to 5 weeks
Bioavailability:
- It has a bioavailability of 89%.
Binding to Albumin:
- Semaglutide is extensively bound to albumin. >99% of the drug is albumin-bound.
The elimination half-life of Semaglutide:
- The elimination half-life of Semaglutide is about one week. The drug can exert its effects for up to five weeks after the last dose of administration.
Metabolism:
- Semaglutide is metabolized by proteolytic degradation and beta-oxidation of the fatty acid chains.
Excretion:
- It is excreted via feces and urine. About 3% of the drug is excreted as such in the urine.
Wegovy Injection Price:
Prices vary in different parts of the world. It is usually advised for patients whose insurance covers the cost. With insurance coverage, the cost can be as low as USD 10.
In other parts of the world, the cost is very variable. It is better to check at your local pharmacy.
In addition, NOVO also provides a patient support program and delivers Ozempic at a reasonable price compared to most local pharmacies.
Wegovy Price in the United States:
Prices vary, but the price of one 2 ml injection pen costs around $1418 USD.
Wegovy Price in Canada:
A 2 ml injection of Wegovy may cost around $1430 USD without insurance.
Wegovy Price in the UK:
Wegovy may cost around £199 per 28 days.
Wegovy Price in Australia:
Wegovy’s monthly cost is around 1627 AUD without insurance.







