Rybelsus, Ozempic, and Wegovy are the three different formulations of Semaglutide. Rybelsus and Ozempic have been approved by the FDA for the treatment of Diabetes, while Wegovy has been approved for the treatment of Obesity.
Because weight loss is one of the intended side effects of all three medications, they are attractive choices for the treatment of prediabetes as well.
Diabetes and Prediabetes Remission after Weight Reduction:
Weight loss of any degree is associated with at least some benefits. However, a weight loss of 5% or more of the initial body weight is associated with significant improvement in the metabolic parameters [Ref]
Metabolic Effects of 5% or more weight loss |
| Improves pancreatic beta-cell function |
| Increase insulin sensitivity (Decrease Insulin Resistance) |
| Reduction in adiposity |
Since weight loss is associated with an improvement in beta-cell function and a reduction in insulin resistance, it has direct effects on blood glucose and A1C.
It is not surprising to see weight loss drugs in clinical trials of diabetes prevention. Among the drugs that have been studied to prevent diabetes, two of them have been FDA-approved for weight loss.
Drugs that may prevent or delay the onset of Diabetes:
- Orlistat (Xenical, Alli)
- Metformin (Glucophage)
- Alpha-glucosidase inhibitors (Acarbose and Miglitol)
- GLP-1 agonists (Liraglutide and the new GLP-1 Semaglutide)
- Thiazolidenediones (Pioglitazone)
Orlistat is a drug purely used for weight loss, while GLP-1 agonists (Liraglutide and Semaglutide) have been approved for the treatment of Diabetes and weight loss.
Metformin is one of the most commonly used drugs for the treatment of prediabetes, however, it is not associated with weight loss. Some weight loss has been mentioned in clinical trials, but it has not been approved for weight loss treatment.
When used to treat prediabetes, metformin solely works to reduce insulin resistance.
Metformin indications for prediabetes:
- BMI >35 kg/m2
- Age less than 60 years
- Women with a history of gestational diabetes
- Rising A1C despite intensive physical therapy
How much A1C reduction is expected with various weight loss Drugs:
Weight loss is associated with an improvement in diabetes control. Drugs that are purely used for treating obesity and weight loss have been observed in various clinical trials to improve A1C by up to 0.5%.
The two FDA-approved weight-loss drugs that have also been approved for the treatment of diabetes have significant effects on A1C.
Liraglutide and Semaglutide improve A1C by approximately 1% to 1.8% in diabetic individuals. Semaglutide (Wegovy), when used in patients with prediabetes, resulted in an A1C reduction of 0.34 relative to placebo.
This 0.34% reduction in A1C converted 84.1% of the prediabetic obese individuals to normal (compared to 47.8% in the placebo group) [Ref].
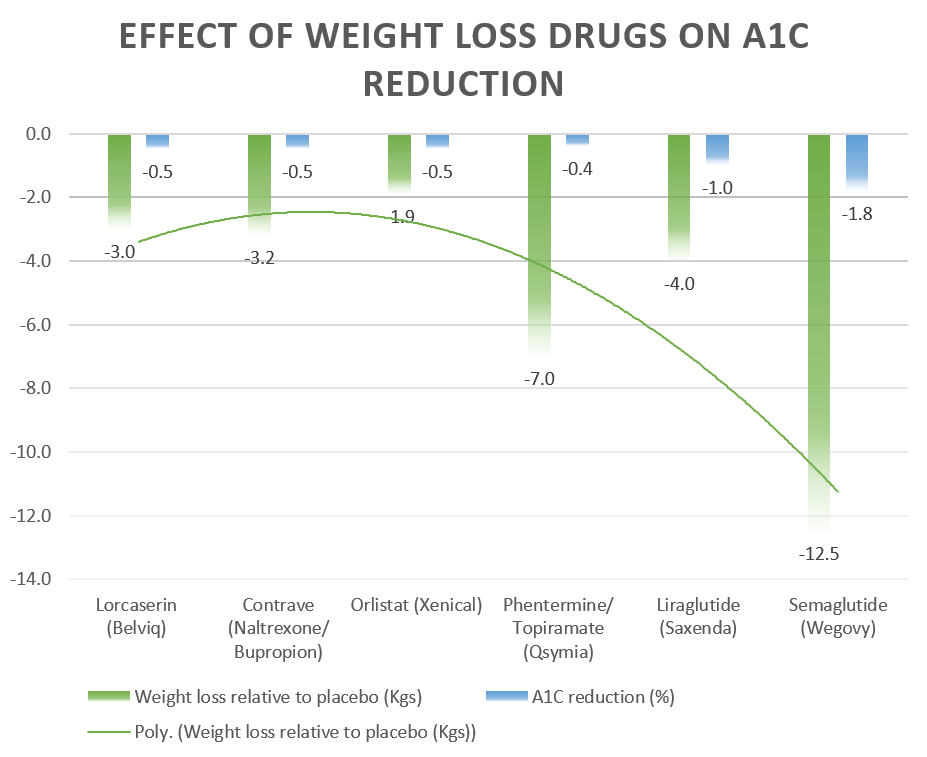
A summary of the weight loss effects and A1C reduction of the six FDA-approved weight-loss drugs is presented in the table below:
FDA-Approved Weight Loss Drugs | Weight loss relative to placebo (Kgs) | A1C reduction |
| Lorcaserin (Belviq) | 3 kgs | -0.5% |
| Contrave (Naltrexone/ Bupropion) | 3.2 kgs | -0.5% |
| Orlistat (Xenical) | 1.9 kgs | -0.5% |
| Phentermine/ Topiramate (Qsymia) | 7 kgs | -0.4% |
| Liraglutide (Saxenda) | 4 kgs | -1.0% |
| Semaglutide (Wegovy) | 12.5 kgs | -0.34% (in Prediabetes)-1.8% with Ozempic in Diabetic patients |
Apart from weight loss drugs, metabolic surgeries have been observed to cause the greatest weight loss in the shortest interval of time.
In addition to weight loss, metabolic surgeries have also been observed to cure diabetes (or “diabetes in remission” as some people don’t like the word “cure diabetes”)
The indications of metabolic surgery include:
- BMI >39 kg/m2 or >37.4 in Asian Americans
- BMI of 35 – 39.9 kg/m2 (32.5 – 37.4 kg/m2 in Asian Americans if hyperglycemia is not adequately controlled with diet, lifestyle intervention, and optimal medical therapy.
- BMI 30 – 34.9 (27.5 – 32.4 in Asian Americans) in patients with diabetes mellitus, despite optimal medical treatment (including oral and injectable)
Rybelsus for Prediabetes:
Rybelsus is an approved medicine for the treatment of Diabetes. So, it is logical to use it in prediabetes, especially for those individuals who have the following features:
- Rising HbA1C despite exercise and a low-calorie diet
- Obese or overweight individuals
- Women with a history of gestational diabetes or secondary diabetes
- Strong family history of diabetes
- Underlying metabolic syndrome or eating disorder
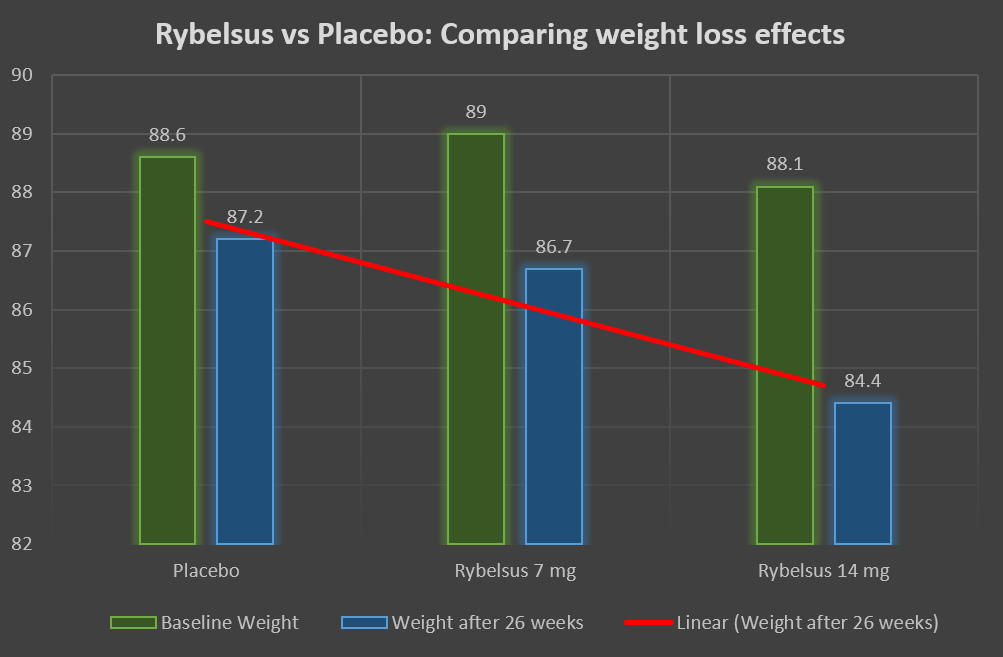
In diabetic individuals, Rybelsus lowers the A1C by 1.3%. However, in prediabetes, the A1C reduction may not be that marked as the baseline A1C is much lower in these individuals.
Even in individuals with an A1C of 6.4%, which is just 0.1% below the diabetic range, Rybelsus may lower it by at least some percentage points.
The combined effects of A1C reduction as a result of weight loss plus the direct A1C-reducing effects of Rybelsus may convert a prediabetic percentage to a totally normal non-diabetic.
Let’s assume that a prediabetic person with a baseline A1C of 6.4% is initiated on Rybelsus treatment.
We can assume that Rybelsus lowers the A1C by at least one-third of that in diabetic individuals (1/3 x -1.3% = -0.4%).
In addition, Rybelsus is associated with a weight loss of 8.1 pounds or 3.6 kg. Like all other weight loss medications, which caused a reduction in the A1C by -0.5%, we expect Rybelsus to lower the A1C by at least -0.4% to -0.5%.
Rybelsus effects on A1C and weight in Diabetic patients | |
| A1C Reduction | -1.3% |
| Weight loss | 8.1 pounds or 3.67 kgs |
Rybelsus effects on A1C and weight in prediabetic patients | |
| A1C Reduction | -0.4% |
| A1C reduction as a result of Weight loss | -0.4% to -0.5% |
The combined weight loss effect plus the direct A1C reduction will result in a cumulative A1C reduction of at least -0.8%.
Hence, a prediabetic individual with an A1C of 6.4% will have an A1C of 5.6% after three to six months, which is the A1C of an average non-diabetic individual.
In conclusion, Rybelsus use for 3 to 6 months may convert a prediabetic person to a totally normal individual. However, these theoretical assumptions must be verified in clinical trials as well.
Ozempic for Prediabetes:
Ozempic is another formulation of Semaglutide that is available as a once-weekly injection. It is primarily approved for the treatment of Diabetes.
Like Rybelsus, Ozempic can have direct effects on the A1C. It may also lower the A1C by reducing body weight, which can have an indirect effect on the A1C.
Since it is a more potent antidiabetic drug compared to Rybelsus, it may lower the A1C rapidly.
In addition, it is associated with significant weight loss, and the indirect A1C-reducing effects of Ozempic will rapidly convert a prediabetic individual into a completely normal non-diabetic individual.
The effect of Ozempic on Weight loss and A1C in Diabetic and Prediabetic individuals:
Ozempic effects on A1C and weight in Diabetic patients | |
| A1C Reduction | -1.2% to -1.8% |
| Weight loss | 13 pounds or 5.89 kgs |
Ozempic effects on A1C and weight in prediabetic patients | |
| A1C Reduction | -0.4% – -0.6% |
| A1C reduction as a result of Weight loss | -0.4% to -0.5% |
From the table above, it can be observed that Ozempic is comparably more potent than Rybelsus as it reduces the A1C by up to 1.8% compared to 1.3% with Rybelsus.
Similarly, it is associated with more weight loss than Rybelsus (13 pounds vs 8 pounds).
It can be estimated that Ozempic can reduce the A1C by at least one-third in prediabetic individuals compared to diabetic individuals.
The cumulative A1C reduction (as a direct effect of Ozempic plus A1C reduction due to weight loss) may be equal to 1%. Reducing HbA1C by 1% can convert a prediabetic individual to a completely normal non-diabetic individual.
Wegovy for Prediabetes:
Wegovy is a high-dose (2.4 mg weekly administered) Semaglutide. It is only approved for the treatment of overweight and obesity.
In clinical trials, it has been observed to cause a weight loss of about 16% from the baseline. This weight loss is usually linked with marked improvement in the symptoms of sleep apnea, hypertension, PCOS, diabetes, and prediabetes.
In clinical trials, Wegovy use was associated with normoglycemia in 84.1% of prediabetic individuals compared to 47.8% in the placebo group.
This means out of 100 prediabetic individuals, Wegovy can convert 84 individuals into normal normoglycemic individuals at week 68.
Unlike, Rybelsus and Ozempic where we theoretically estimated the potential of the two drugs to convert prediabetic individuals to normoglycemic ones, Wegovy’s use in prediabetic patients is evidence-based [Ref].
- Premium Ingredients: Our tea blend features high-quality Berberine, aromatic Oolong, and refreshing Mint, all organicall…
- Mint Flavored: Enjoy the cool, crisp taste of mint that perfectly complements the robust flavors of Berberine and Oolong…
- Unsweetened with Zero Calories: Crafted for those who appreciate the natural flavors of tea, our blend is completely uns…
- CLINICAL-GRADE KETONE & GLUCOSE TESTING The GK+ Meter delivers fast, accurate readings for both ketones and glucose. Tru…
- COMPLETE STARTER KIT Includes everything you need to start testing: GK+ Bluetooth meter, 10 ketone strips, 10 glucose st…
- EFFORTLESS APP INTEGRATION FOR SMARTER TRACKING Automatically sync your results to the free MyMojoHealth app to view tre…






