Dupilumab (Dupixent) is a biological medicine that got FDA approval in 2017 for the treatment of allergic asthma, atopic dermatitis, and chronic rhinosinusitis with nasal polyposis.
Dupilumab is a human monoclonal antibody that is prepared by recombinant DNA technology in Chinese Hamster ovary cell suspension culture.
It is a human monoclonal antibody of the IgG4 subclass that binds to the IL-4Rα and inhibits the signaling of IL-4 and IL-13.
IL-4 is a cytokine that is produced primarily by mast cells, eosinophils, basophils, and Th2 cells. It differentiates Th cells into Th2 cells.
It plays an important role in the pathogenesis of autoimmune and allergic conditions such as asthma and atopic dermatitis.
IL-13 has structural and functional similarities to IL-4. It is active in cells that are infested by parasitic infections and help them clear from the body.
It has a central role in inflammatory allergic conditions throughout the body.
Dupilumab (Dupixent) Uses (Indications):
Dupilumab (Dupixent) is used to treat the following conditions [Ref]:
Atopic Dermatitis:
It is indicated for the treatment of children who are 6 years of age or above and have moderate to severe atopic dermatitis that is not responding to the conventional treatment.
It can be used with or without topical corticosteroid therapy.
Atopic dermatitis is also called Eczema. It is a chronic condition with frequent flares and may or may not be associated with allergic asthma.
Patients with Eczema or atopic dermatitis have localized areas of skin inflammation (dermatitis). The skin is itchy, red, and serous fluid may be oozing out. If left untreated, the skin becomes thickened and scaly.
Atopic dermatitis is not curable but treatable. Dupilumab use in atopic dermatitis resulted in marked symptomatic improvement in most of the clinical symptoms.
Asthma:
Dupilumab (Dupixent) is approved for the treatment of children who are 12 years of age or above and have moderate to severe eosinophilic or allergic asthma or those who have oral corticosteroid-dependent asthma.
Eosinophilic or allergic asthma is a variant of asthma that affects people who have a personal or a family history of atopy or allergies. Atopic conditions other than asthma include eczema or atopic dermatitis, rhinosinusitis, and food allergies.
Patients with eosinophilic asthma usually respond to inhalational or oral corticosteroids. However, those with moderate to severe asthma who have poor symptom control may be treated with biologics such as Dupilumab (Dupixent).
Chronic Rhinosinusitis with nasal polyposis (CRwNP):
It is indicated as an add-on maintenance treatment for adults with chronic rhinosinusitis with nasal polyposis who are inadequately controlled with conventional treatment.
Eosinophilic Esophagitis:
The FDA has recently approved Dupilumab (Dupixent) as a first-in-class medicine for the treatment of adults and children above 12 years of age with eosinophilic esophagitis who weigh at least 40 kgs.
Patients with eosinophilic esophagitis have dysphagia, heartburn, and pain in the epigastrium. It is characterized by inflammation and eosinophilic infiltration into the esophageal mucosa.
Dupilumab (Dupixent) Dosage:
Dupixent is available as:
- Prefilled Pen, and
- Prefilled syringe
Administering the dose via the prefilled pen is only intended for adults and children who are 12 years of age or older.
The dose in adults who are 18 years of age or older is fixed, however, in children 6 to 17 years of age, the dose is based on body weight.
Dosing Recommendations of Dupixent in Patients with Atopic Dermatitis:
Initial Dose | Maintenance Dose | |
Adults | 600 mg (2 injections of 300 mg per injection) | 300 mg every other week |
Children (6 – 17 years of age): Dose based on body weight | ||
| 15 to 29 kg | 600 (2 injections of 300 mg per injection) | 300 mg monthly (every four weeks) |
| 30 to 59 kg | 400 (2 injections of 200 mg per injection) | 200 mg every other week |
| 60 or more kgs | 600 (2 injections of 300 mg per injection) | 300 mg every other week |
In addition to Dupilumab (Dupixent), patients may use topical corticosteroids and Calcineurin inhibitors such as Pimecrolimus (Elidel).
The use of Calcineurin inhibitors (Pimecrolimus or Elidel) should be reserved for skin areas that are hard to treat. These may include the skin of the face, genitals, axilla (armpits), and groin.
Dupilumab (Dupixent) Dose in Asthma:
Dupilumab (Dupixent) is indicated only for the treatment of moderate to severe allergic (eosinophilic asthma in adults and children who are at least 12 years of age.
The following table simplifies the dosing schedule of Dupixent in patients with Asthma:
Eosinophilic or Allergic Asthma | |
| Initial Dose | 400 (2 injections of 200 mg per injection) |
| Maintenance Dose | 200 mg every other week |
Oral Corticosteroid-dependent asthma or Asthma with co-morbid moderate to severe atopic dermatitis for which Dupixent may be indicated | |
| Initial Dose | 600 (2 injections of 300 mg per injection) |
| Maintenance Dose | 300 mg monthly (every four weeks) |
Dupilumab (Dupixent) Dose in Chronic Rhinosinusitis with Nasal Polyposis:
It is recommended in a dose of 300 mg every other week in adults.
Dupilumab (Dupixent) Dose in Eosinophilic Esophagitis:
In patients with eosinophilic esophagitis, Dupixent is given in a dose of 300 mg every week [Ref]
Dupixent Dosage Forms and Strengths:
Dupixent is available as:
Dupixent Pen |
| 300 mg per 2 ml in a single prefilled pen |
| 200 mg per 1.14 ml in a single prefilled pen |
Dupixent Syringe |
| 300 mg per 2 ml in a single-dose prefilled syringe with a needle shield |
| 200 mg per 1.14 ml in a single-dose prefilled syringe with a needle shield |
How to administer Dupilumab (Dupixent)?
Dupixent prefilled pen is a one-time disposable pen. It is administered subcutaneously into the skin of the front of the abdomen 3 to 5 cm away from the navel, the thighs, and the upper arms.

The skin of the upper arms should only be used by a healthcare provider. Patients should avoid injecting it into the upper arms.
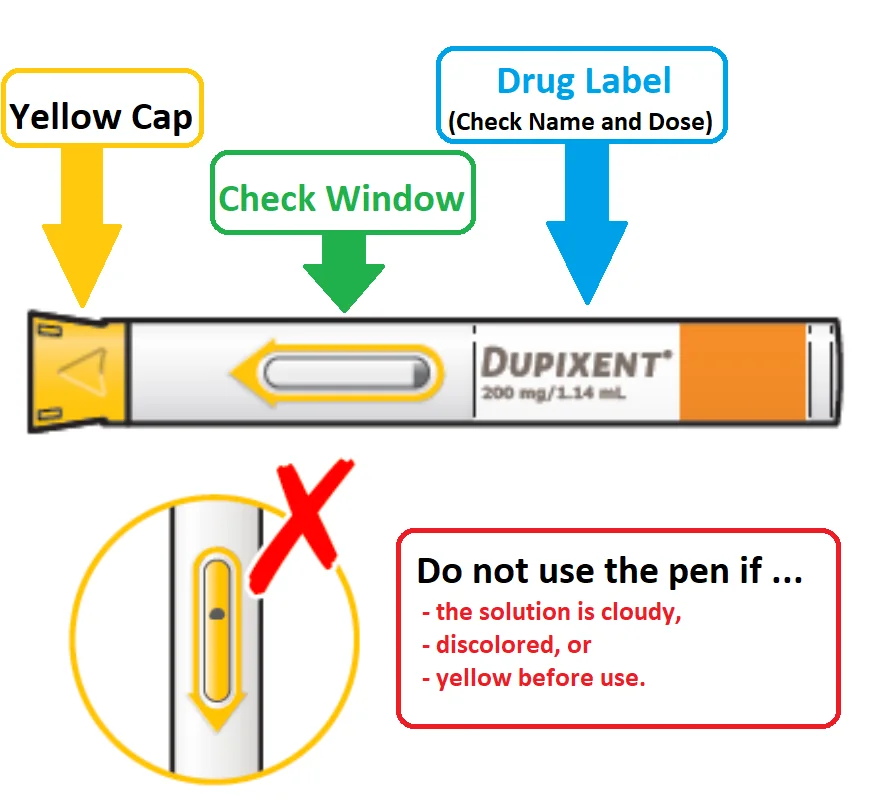
Before injecting, check the following:
- See the expiry date. If the date of expiry has passed, do not use it
- Inspect the solution by looking into the “check Window”. If the solution is cloudy or discolored, do not use it.
- If the Check window is yellow before use, do not use it.
- Check the label for the proper dose and name.
After checking the pen, keep it at room temperature (not exceeding 25 C or 77 F) for 30 minutes. Avoid heating the injection or keeping it under hot water or in a microwave.
Clean the skin with an alcohol swab and let it dry. Take the pen, keep it straight (at 90 degrees to the skin) and press it firmly against the skin.

The orange needle cover will disappear and a click sound will be heard. Simultaneously, the “check window” will start to become yellow.
Once all the dose is injected, the “check window” will be totally yellow, and a second “click” will be heard.
After the second click, wait for five seconds or count up to five, and then remove the pen. The orange cover will reappear.

If the “check window” is partially yellow or medicine is coming out of the needle, it means the injection has not been fully administered.
You may consult your healthcare provider but do not administer another dose.
Dupixent doses of 400 mg and 600 mg should be administered as two separate injections of 200 mg and 300 mg each. The site of the two injections should not be the same.
Also, avoid injecting into the skin that is red, tender, bruised, or has induration. The injection sites should be rotated each time the injection is administered.
If the dose is missed, administer it as soon as possible. If the dose has been missed for less than 7 days of the scheduled dose, administer the dose and follow the old schedule.
However, if more than 7 days have passed, the dose should be administered and the following dose should be scheduled from the date of the current injection.
Dupilumab (Dupixent) Dose in Kidney and Liver Diseases:
The manufacturer has not recommended any dosage adjustments in patients who have underlying liver or kidney diseases.
Dupilumab (Dupixent) Dose in Pregnancy and Breastfeeding:
Dupixent has not been studied in pregnant and nursing mothers. Because Dupxient is a subclass of IgG4, it can cross the placental barrier as well as it can be excreted in breast milk.
Since studies have not been conducted, it is advisable to avoid Dupixent in pregnant mothers. In lactating mothers, the risks-to-benefit ratio may be taken into consideration.
Dupilumab Drug Interactions:
Live Vaccines:
Avoid administering live vaccines while being treated with Dupixent.
Non-live vaccines:
Dupixent has been studied in patients who were administered polysaccharide vaccines (Tdap and meningococcal vaccines).
Dupixent did not interact with the vaccines. Antibody titers after vaccination were similar in both the Dupilumab-treated group and the placebo-treated group.
Belimumab:
The toxic effects of belimumab may be increased.
Dupilumab (Dupixent) Contraindications:
Dupilumab (Dupixent) should be avoided in patients who are allergic to the drug or any of its components.
Allergic reactions may be severe and result in facial swelling, hypotension, and bronchospasm. The first dose should always be administered in a hospital setting where measures to resuscitate the patient are adequate.
In clinical trials, hypersensitivity reactions occurred in less than 1% of the patients. These reactions included:
- Rash
- Urticaria
- Erythema Nodosum
- Serum sickness or serum sickness-like reactions, and
- Anaphylaxis
Dupilumab (Dupixent) Warnings & Precautions:
The incidence of side effects of Dupixent, as reported in clinical trials, were different in frequency when patients were treated for asthma vs atopic dermatitis vs chronic rhinosinusitis with nasal polyposis.
Before enlisting the common side effects, it is important to read a few warnings and precautions about the following conditions:
Keratoconjunctivitis:
Keratitis and conjunctivitis (keratoconjunctivitis) occurred in patients who were treated with Dupixent.
It manifests as watery eyes, redness, photosensitivity, and pain. The condition settles on its own within the first week, however, consultation with an eye specialist may be recommended if the symptoms are severe.
The incidence of developing keratoconjunctivitis was more common in patients who were treated with Dupixent for atopic dermatitis (10% vs 2%), followed by those treated for chronic rhinosinusitis with nasal polyposis (2% vs 1%).
The incidence of keratoconjunctivitis was seen in similar frequency in patients treated for asthma with Dupixent vs Placebo.
Eosinophilic Conditions:
It is not uncommon for asthmatic patients or those with chronic rhinosinusitis to have an underlying vasculitis or other eosinophilic syndromes such as eosinophilic pneumonia and eosinophilic granulomatosis with polyangiitis.
Since treatment with Dupixent is associated with a reduction in the requirements of oral corticosteroids, healthcare providers must be vigilant in diagnosing these rare conditions.
If the patient’s clinical condition deteriorates with Dupixent therapy after corticosteroids were tapered off or withdrawn, always investigate the patient for other systemic hypereosinophilic syndromes.
Effect on acute symptoms of asthma:
Dupixent is not indicated for the treatment of acute severe asthma or acute exacerbations of asthma.
In patients treated with Dupixent, if the clinical condition of the patient deteriorates, treatment must be discontinued and appropriate management including oral or injectable corticosteroids should be administered.
In addition, for patients who are being treated with Dupixent for atopic dermatitis or chronic rhinosinusitis with nasal polyposis and have co-morbid asthma, it is not recommended to discontinue asthma medications without first consulting your healthcare provider.
Parasitic infections:
Before initiating Dupixent therapy, investigate and treat patients for helminthic infections. Treatment with Dupixent may impair the body’s immune response to helminthic infections.
In addition, if a person develops any new onset of helminthic infections while being treated with Dupixent, Dupixent therapy may be discontinued especially if the helminthic infection is non-responsive to therapy.
Adjusting Corticosteroids dose:
Do not abruptly discontinue corticosteroids when treatment with Dupixent is initiated. Corticosteroids should be tapered off gradually.
While tapering corticosteroids, monitor patients for the emergence of new symptoms that might indicate an underlying hypereosinophilic syndrome, like eosinophilic pneumonia and eosinophilic granulomatosis with polyangiitis.
Side effects of Dupixent:
The frequency of side effects differed in patients treated for asthma, atopic dermatitis, and chronic rhinosinusitis.
Side effects of Dupixent in patients treated for atopic dermatitis:
The most common side effects that were present in 1% or more of the patients treated with Dupixent were:
- Injection site reactions
- Conjunctivitis
- Blepharitis
- Oral herpes
- Keratitis
- Eye Pruritis
- Other herpes simplex infections
- Dry eyes
Side effects of Dupixent in patients treated for Asthma:
The most common side effects in Dupixent-treated asthma patients included:
- Injection-site reactions
- Oropharyngeal pain
- Eosinophilia
Side effects of Dupixent in patients treated for chronic rhinosinusitis:
The most common side effects encountered in patients treated with Dupixent who had chronic rhinosinusitis were:
- Injection-site reactions
- Conjunctivitis
- Arthralgia
- Gastritis
- Insomnia
- Eosinophilia
- Toothache
In addition to the above side effects, patients treated with Dupixent may develop antibodies directed against the drug. The clinical implications of these antibodies are not yet clear.
Dupilumab (Dupixent) Monitoring Parameters:
Monitor the patients for evidence of any hypersensitivity reactions, especially after the first dose.
Patients with asthma should have their pulmonary functions done periodically to see for treatment response.
Monitor all patients for signs and symptoms of infections. Specifically, monitor patients for evidence of parasitic and helminthic infections.
Dupilumab (Dupixent) MOA (Mechanism of action:
Dupilumab (Dupixent) is a human monoclonal antibody of the IgG4 subclass. It specifically targets the IL-4Rα receptor complex that is shared by both IL-4 and IL-13.
Thus, by binding to IL-4Rα, it inhibits the signaling pathway executed by both IL-4 and IL-13. Both these cytokines help in the differentiating of Th2 cells and are released primarily by eosinophils, basophils, and mast cells.
The IL-4 receptor signaling is inhibited by type-1 receptors while the type 2 receptors block the signaling of both IL-4 and IL-13.
The end result of blocking these two cytokines is a reduction in the release of important inflammatory mediators that are involved in the pathogenesis of asthma, atopic dermatitis, eosinophilic esophagitis, and chronic rhinosinusitis with nasal polyposis.
These inflammatory mediators include:
- Histamine
- Leukotrienes
- Eicosanoids
- Cytokines
- Chemokines
- Nitric oxide, and
- IgE
Bioavailability after subcutaneous administration:
- 61 to 64%
The time to peak plasma concentration:
- One week
The time to reach steady-state concentration:
- 16 weeks
Metabolism:
- Like all other antibodies, Dupixent is degraded by proteolytic enzymes into amino acids that are utilized inside the body.
- Complete clearance of the drug from the plasma (after discontinuation) ranges from 9 to 13 weeks.
Dupilumab Brands and Price:
Dupilumab is only available by the trade name Dupixent.
Dupixent is very costly, however, it is covered by insurance in most cases.
Copay cards and discounts are also available once your healthcare provider confirms your eligibility for the injection.
The per carton (monthly dose) of Dupixent is USD 3384.83 [Ref].
One single injection costs USD 1692.415







