Brand Name: Tzield
Drug Name: Teplizumab-mzwv
Manufacturer’s Name: Provention Bio Inc
FDA Approval Year: 2022 [Ref]
Primary Indication: Prevention of Type 1 Diabetes
Tzield (Teplizumab-mzwv) injection is a monoclonal antibody (of the IgG class). It is directed against CD3-positive T-cells preventing the death of islet cells in the pancreas.
It has been approved by the FDA for the prevention of Type 1 Diabetes in adults and children older than 8 years of age who are at risk.
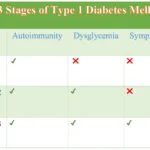
Type 1 diabetes is classified into three different stages based on the:
- Presence of autoantibodies
- Dysglycemia, and
- Symptoms of diabetes
The three stages of Type 1 Diabetes are summarized in the table below:
The 3 Stages of Type 1 Diabetes Mellitus | |||
| Autoimmunity | Dysglycemia | Symptomatic | |
| Stage 1 | ✔ | ❌ | ❌ |
| Stage 2 | ✔ | ✔ | ❌ |
| Stage 3 | ✔ | ✔ | ✔ |
Individuals who have been diagnosed with Stage 2 Type 1 Diabetes are candidates for receiving Tzield (Teplizumab-mzwv).
It is important to exclude patients with Type 2 Diabetes before treatment initiation.
| You may also like to read: |
Tzield (Teplizumab-mzwv) Dose:
Before initiating TZIELD treatment, patients should undergo laboratory evaluations and vaccinations.
The following conditions should be excluded before starting TZIELD:
Parameters | Cut-off |
| Lymphocyte count | < 1,000 lymphocytes/mcL |
| Hemoglobin | < 10 g/dL |
| Platelets | < 150,000/mcL |
| Absolute neutrophil count | < 1,500 /mcL |
| Liver functions | ALT or AST: > 2 times the upper limit of normal or Bilirubin: > 1.5 times the ULN |
| Laboratory | Acute EBV or CMV infections |
| Infections | Active severe infection or chronic ongoing infection except for mild localized skin infections |
Dilute the injection and pre-medicate the patient before the infusion with an NSAID or acetaminophen, an antihistamine, and an antiemetic for the first 5 days of the dose.
The recommended dosage of TZIELD for administration is as follows:
It is administered intravenously once daily for 14 consecutive days, based on body surface area dosing:
- Day 1: 65 mcg/m²
- Day 2: 125 mcg/m²
- Day 3: 250 mcg/m²
- Day 4: 500 mcg/m²
- Days 5 through 14: 1,030 mcg/m²
Avoid giving two doses on the same day.
Additional Preparation and Administration Instructions
The following instructions must be followed for additional preparation and administration of TZIELD:
- Visually inspect TZIELD before use.
- Use an aseptic technique to prepare TZIELD.
- Each vial of TZIELD is intended for single-dose use only.
- Dilute the medication with normal saline to make the final solution of 100 mcg/ml and then withdraw the required dose.
- Slowly add the TZIELD dose from the syringe to a 25 mL saline infusion bag.
- Discard any unused solution. Store the infusion at room temperature if the infusion is delayed. Infuse within 2 hours after preparing the solution.
- If it is not administered within 4 hours, discard it.
| You may also like to read: |
Teplizumab Contraindications:
No contraindications are given in the FDA prescribing information. However, one should take precautions in the following conditions:
Cytokine Release Syndrome (CRS):
TZIELD treatment can cause cytokine release syndrome (CRS) which was seen in 5% of patients in clinical trials.
Symptoms of CRS include fever, nausea, fatigue, headache, muscle pain, joint pain, and higher liver enzyme levels.
To reduce the risk of CRS, healthcare providers should follow these steps:
- Administer antipyretics, antihistamines, and/or antiemetics before the treatment
- Monitor liver functions. Stop TZIELD treatment if the ALT or AST exceeds 5 times the ULN or if the bilirubin exceeds 3 times the ULN.
- If severe CRS develops, pause dosing for 1-2 days and then continue the remaining doses to finish the 14-day course on consecutive days, or stop treatment altogether.
Serious Infections
Since the drug inhibits the functioning of T lymphocytes, severe bacterial and viral infections may occur.
Most infections in the clinical trials reported were: gastroenteritis, cellulitis, pneumonia, abscess, and sepsis. The treatment should be deferred in cases of severe active or chronic infections.
Lymphopenia
Lymphopenia is a common side effect of the treatment. It has been reported to affect 78% of the individuals receiving the treatment.
Once the treatment is stopped, the lymphocyte counts rise to normal within 2 weeks.
Monitoring for lymphopenia is recommended. In patients with a lymphocyte count of <500 cells per mcL lasting 1 week or longer, the treatment should be discontinued
Hypersensitivity Reactions:
Allergic reactions including serum sickness, angioedema, urticaria, rash, vomiting, and bronchospasm, have been reported with the treatment.
In severe cases, the treatment should be discontinued.
Vaccinations:
Tzield may reduce the efficacy of vaccines. All individuals should have their age-appropriate vaccination completed before Tzield treatment initiation.
Avoid inactivated or mRNA vaccinations 2 weeks before, during, and 6 weeks after the treatment.
Avoid live-attenuated vaccinations 8 weeks before, during, and 52 weeks after the treatment is completed.
| You may also like to read: |
Tzield (Teplizumab-mzwv) Side effects:
| Side Effect | Incidence Rate | Potential Consequences |
| Cytokine release syndrome (CRS) | 5% | Fever, fatigue, nausea, headache, myalgia and arthralgia, elevated liver enzymes |
| Lymphopenia | 78% | Usually resolves within 14 days and is mild but severe lymphopenia lasting 1 week or longer has been reported. |
| Bacterial and viral infections | 3.5% | Serious infections may occur. Commonly, gastroenteritis, cellulitis, pneumonia, abscess, and sepsis have been reported. |
| Hypersensitivity reactions | Not specified | Serum sickness, angioedema, urticaria, rash, vomiting, and bronchospasm |
| Interference with vaccinations | Not specified | Decrease in vaccine efficacy |
| You may also like to read: |
Use In Pregnancy and Lactation:
The pregnancy risk summary for TZIELD, a monoclonal antibody, is based on limited case reports from clinical trials and indicates that the drug may be associated with risks of major birth defects, miscarriage, and immunosuppression in the fetus.
To minimize fetal exposure, avoid using it during pregnancy and at least 30 days before a planned pregnancy.
The effects of TZIELD on breastfed infants and milk production are unknown. Lactating women may discard breast milk during treatment and 20 days after the treatment to minimize drug exposure to a breastfed child.
| You may also like to read: |
Mechanism of Action of Teplizumab:
Teplizumab is a monoclonal antibody of the IgG class that binds to CD3, a cell surface antigen on T lymphocytes.
It contributes to partial agonistic signaling and deactivation of pancreatic beta cell autoreactive T lymphocytes. It increases the proportion of regulatory T cells and exhausted CD8+ T cells in peripheral blood.
It prevents beta-cell death from autoimmunity and thus prevents the progression of Stage 2 Type 1 Diabetes to Stage 3.
Pharmacokinetics of Teplizumab-mzwv:
Pharmacokinetics | Description |
| Steady State Concentrations | Not achieved during the 14-day infusion. |
| Distribution | The drug distribution is limited to the vascular compartment |
| Elimination | The mean terminal half-life is 4.5 days. |
| Metabolism | It is metabolized into small peptides by catabolic pathways; Metabolites are not expected to have significant pharmacological activity |
| You may also like to read: |
Patient Counseling Information for TZIELD (Teplizumab-mzwv)
Before starting treatment, patients should be advised to read the FDA-approved patient labeling (Medication Guide).
Cytokine Release Syndrome
- Patients should be informed about the signs and symptoms of cytokine release syndrome (CRS), such as fever, chills, and flu-like symptoms.
- If they experience any of these symptoms, they should contact their healthcare provider immediately.
Serious Infections
- Patients should be informed that TZIELD may lower their ability to fight infections.
- If they develop any symptoms of infection, they should contact their healthcare provider.
Lymphopenia
- Most patients experience mild lymphopenia, but a few may develop severe lymphopenia that requires stopping treatment.
- Patients should be informed about this potential side effect.
Hypersensitivity Reactions
- Patients should be advised of the symptoms of hypersensitivity reactions, such as rash, itching, and difficulty breathing.
- They should stop taking it and seek medical attention promptly if they experience any of these symptoms.
Vaccinations
- Patients should complete their vaccinations before starting the treatment and avoid concurrent use of live, inactivated, and mRNA vaccines.
Pregnancy
- Patients should inform their prescriber if they are pregnant, planning to become pregnant or suspect they may be pregnant.
- If exposed to TZIELD during pregnancy, patients should contact Provention Bio, Inc.’s Adverse Event reporting line at 1-844-778-2246.
Lactation
- Lactating women should be advised not to breastfeed their child while on treatment and for 20 days after the last infusion.
| You may also like to read: |