Empagliflozin is one of the potent SGLT2 Inhibitors. Empagliflozin (Jardiance) is the second most potent SGLT2 Inhibitor after Canagliflozin (Invokana).
It has similar glycemic efficacy as Ertugliflozin (Steglatro) and is superior to Farxiga (Dapagliflozin) in lowering blood glucose.
It has proven benefits in the treatment of the following conditions:
- It is approved for the treatment of diabetes mellitus type 2 as an adjunct to lifestyle intervention and exercise as monotherapy or in combination with other antidiabetic medications.
- It has proven efficacy in patients with heart failure with either reduced ejection fraction or preserved ejection fraction. It reduces the risks of hospitalizations due to heart failure.
- It has established efficacy to lower the chances of deaths that occur due to a heart-related condition in patients with established cardiovascular disease.
Empagliflozin is usually given in a dose of 10 mg once daily and the dose is then increased as per the patient’s tolerance to 25 mg (which is the maximum dose).
The original drug has been marketed under the brand name Jardiance. It is also available in combination as:
- Synjardy: It is a combination of metformin and Jardiance
- Glyxambi: It is a combination of Linagliptin and Jardiance
- TriJardy XR: It is a combination of Metformin, Linagliptin, and Jardiance
Common Side effects of the Jardiance (Empagliflozin) include:
- Urinary tract infections
- Hypotension (low blood pressure)
- Acute Renal failure due to dehydration
Empagliflozin MOA (Jardiance Mechanism of Action):
Empagliflozin (Jardiance) is a Sodium-Glucose Co-Transporter 2 Inhibitor (SGLT2). It has minimal effects on other SGLT receptors and is 5000 times more specific for inhibiting SGLT2 receptors compared to other SGLT receptors.
Since SGLT2 receptors are primarily located in the renal tubules, Empagliflozin acts on these receptors and inhibits the reabsorption of filtered glucose.
Thus, glucose that has been filtered into the urine does not get reabsorbed back into the blood.
With more glucose excreted in the urine, more water goes with the glucose. This is called osmotic diuresis. Thus, empagliflozin causes the body to lose glucose, water, and sodium.
Apart from its glucose-lowering effects in patients with diabetes, it is associated with fluid loss and hence beneficial in fluid-overloaded states like heart failure.
The loss of sodium and fluid also helps lower blood pressure. Furthermore, it can cause some degree of weight loss. The initial weight loss associated with its use can be the result of the fluid excreted via the renal tubules.
However, as glucose reabsorption is inhibited, it also causes the loss of calories. In clinical trials, SGLT2 Inhibitors including Empagliflozin (Jardiance) have been observed to cause significant weight loss compared to a placebo.
Empagliflozin Uses:
Empagliflozin (Jardiance) is used to treat patients with diabetes and has currently been approved for the treatment of other conditions in diabetic and non-diabetic individuals.
Empagliflozin efficacy in the treatment of T2DM:
The efficacy of empagliflozin has been studied in patients with diabetes mellitus. More than 900 diabetic patients who had uncontrolled diabetes with glycated hemoglobin between 7 to 10% were randomized into three groups:
- One group of patients received a placebo medicine
- Another group of patients received Empaliflozon in a dose of 10 mg once daily
- The third group of patients received the drug at a dose of 25 mg once daily.
Jardiance was significantly more effective compared to a placebo in diabetic patients when used as monotherapy.
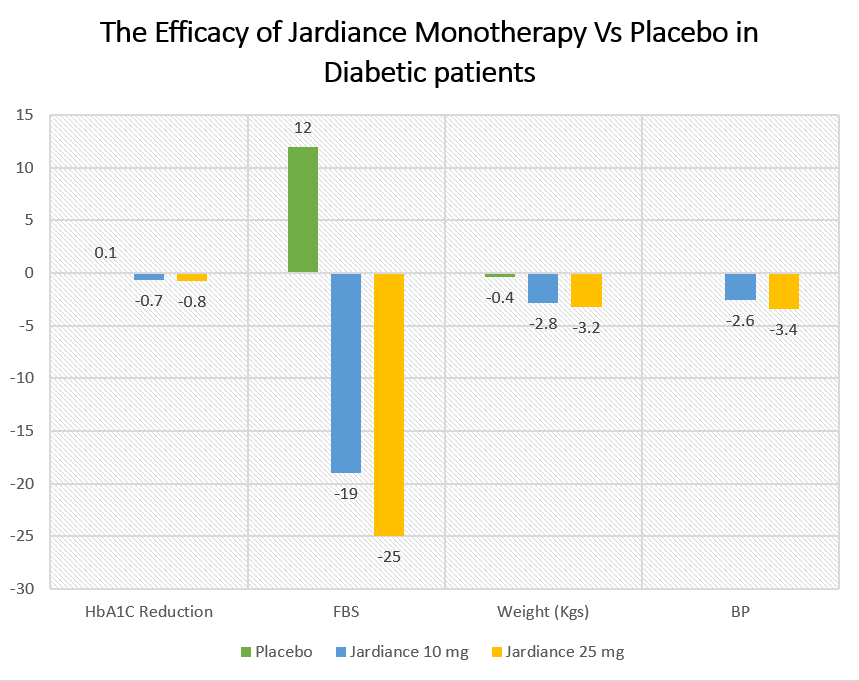
The effect on Fasting Blood Glucose, Glycated hemoglobin, Weight, and blood pressure after 24 weeks was as:
Placebo | Jardiance 10 mg | Jardiance 25 mg | |
| HbA1C Reduction | 0.1 | -0.7 | -0.8 |
| FBS | 12 | -19 | -25 |
| Weight (Kgs) | -0.4 | -2.8 | -3.2 |
| BP | -2.6 | -3.4 |
The efficacy of Synjardy (Empagliflozin and Metformin Combination) in patients with T2DM:
Synjardy is a combination of Metformin and Empagliflozin. In clinical trials, Synjardy has been found to be more effective in lowering the glycated hemoglobin after 24 weeks compared to either drug alone.
The results of the study are presented here in the table given below:
Drug Treatment | HbA1C Reduction |
| Jardiance 10 mg | -1.4 |
| Jardiance 25 mg | -1.4 |
| Metformin 1000 mg | -1.2 |
| Metformin 2000 mg | -1.8 |
| Jardiance 10 mg + Metformin 1000 mg | -2 |
| Jardiance 10 mg + Metformin 2000 mg | -2.1 |
| Jardiance 25 mg + Metformin 1000 mg | -1.9 |
| Jardiance 25 mg + Metformin 2000 mg | -2.1 |
The efficacy of Empagliflozin added to Metformin and Sulfonylurea in patients with uncontrolled T2DM:
Empagliflozin was studied in patients with uncontrolled diabetes who were already on metformin or sulfonylurea. Patients were divided into three groups:
- One group of patients received empagliflozin in a dose of 10 mg in addition to metformin and sulfonylurea
- The other group received empagliflozin in a dose of 25 mg along with metformin and sulfonylurea
- The third group received a placebo medicine and continued on metformin plus Sulfonylurea.
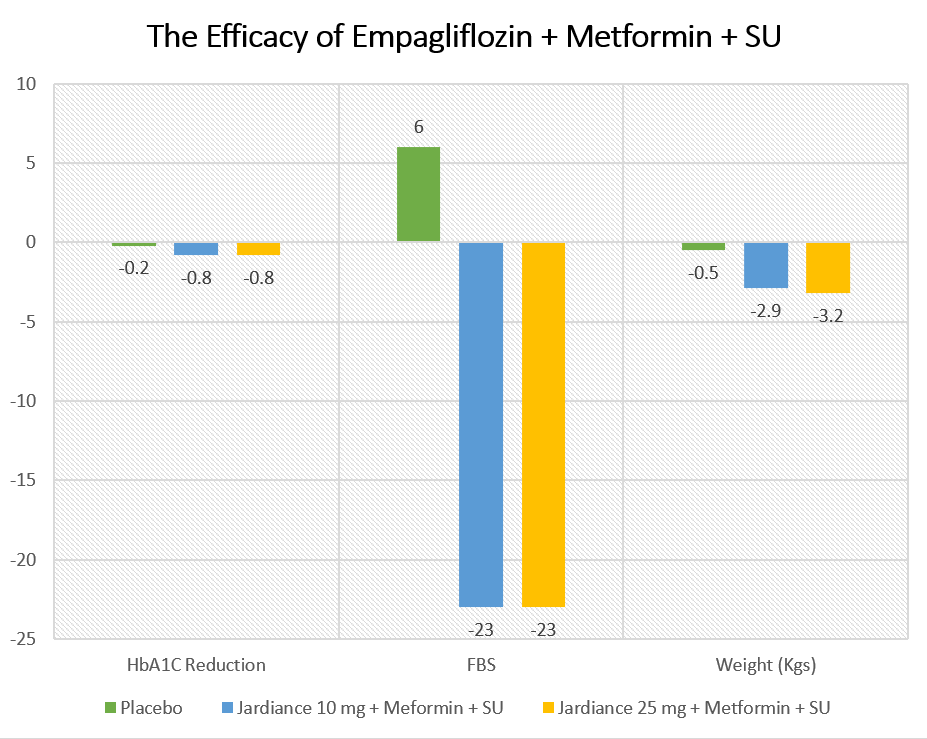
The results of the study are summarized in the table below:
| Placebo | Jardiance 10 mg + Meformin + SU | Jardiance 25 mg + Metformin + SU | |
| HbA1C Reduction | -0.2 | -0.8 | -0.8 |
| FBS | 6 | -23 | -23 |
| Weight (Kgs) | -0.5 | -2.9 | -3.2 |
Empagliflozin has also been found to be superior when given to diabetic patients in combination with Linagliptin (Tradjenta) and metformin compared to either drug alone.
Jardiance was given in a dose of either 10 mg or 25 mg while the dose of Linagliptin was fixed at 5 mg.
Jardiance direct comparison with Glimepiride (Amaryl):
Jardiance has been compared directly with Glimepiride. Jardiance was given in a fixed dose of 25 mg along with metformin to one group of patients.
The other group of patients received metformin and glimepiride mean dose of 2.7 mg (the maximum dose is 8 mg).
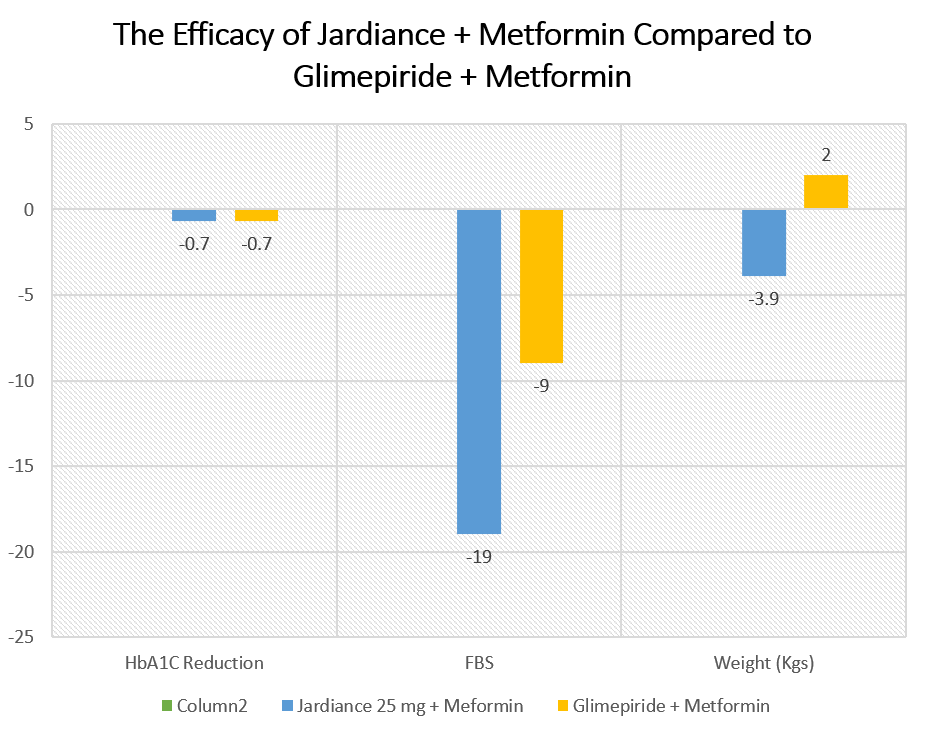
The results of the study are given here in the table below:
| Jardiance 25 mg + Meformin | Glimepiride + Metformin | |
| HbA1C Reduction | -0.7 | -0.7 |
| FBS | -19 | -9 |
| Weight (Kgs) | -3.9 | 2 |
Jardiance in Comparison with Pioglitazone:
Jardiance was also compared to pioglitazone in one study. Patients who were on pioglitazone and metformin and had glycated hemoglobin ranging from 7 to 10% were enrolled in the study.
- One group of patients received a placebo, pioglitazone, and metformin,
- another group received pioglitazone, metformin, and 10 mg Jardiance, and
- the third group of patients received pioglitazone, metformin, and Jardiance 25 mg.
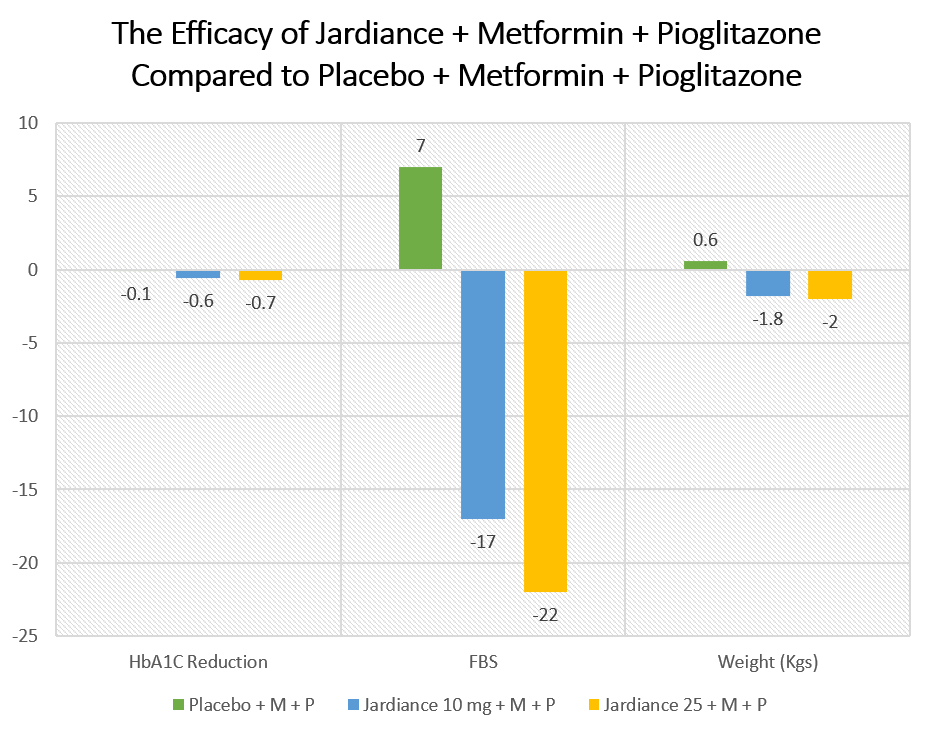
Patients who received Jardiance had significantly better glycemic control compared to placebo. The results were also significant in terms of weight loss and fasting blood glucose. The results of the study are summarized here:
| Placebo + Metformin + Pioglitazone | Jardiance 10 mg + Metformin + Pioglitazone | Jardiance 25 mg + Metformin + Pioglitazone | |
| HbA1C Reduction | -0.1 | -0.6 | -0.7 |
| FBS | +7 | -17 | -22 |
| Weight (Kgs) | +0.6 | -1.8 | -2 |
Similarly, when added to basal insulin or premixed insulin, Jardiance resulted in significant improvement in the glycated hemoglobin compared to placebo.
Empagliflozin efficacy in the treatment of Heart failure:
The EMPA-REG outcome Trial evaluated the safety and efficacy of Jardiance in patients with cardiovascular disease. More than 7000 patients with a mean HbA1C of 8.1% were randomized into three groups and followed for a mean of 3.1 years.
Patients either received a placebo, Jardiance 10 mg, or Jardiance 25 mg daily. More than half (57%) of the patients had diabetes for more than 10 years. About a third of the study patients had microvascular complications.
All study patients had a stable baseline atherosclerotic cardiovascular disease. 76% had coronary artery disease, 23% had a stroke, and 21% had a peripheral arterial disease.
- Jardiance significantly reduced the number of patients dying from cardiovascular disease, stroke, or myocardial infarction.
- Jardiance also reduced the incidence of non-fatal myocardial infarction and cardiovascular deaths
- However, the incidence of stroke increased in patients treated with Jardiance compare to placebo.
In the Emperor-Reduced and Emperor-Preserved Trials, Jardiance has been shown to significantly lower the risks of hospitalizations due to heart failure in patients with HFrEF (heart failure with reduced ejection fraction and HFpEF (Heart Failure with preserved ejection fraction [Ref].
The dose of Jardiance in patients with heart failure or without diabetes is generally lower than that used for diabetes. It is given in a dose of 10 mg once daily.
Lower doses of 5 mg and even 2.5 mg are currently being formulated for use in patients with heart diseases without diabetes.
Empagliflozin efficacy in the treatment of diabetic nephropathy:
Jardiance has been studied in mild, moderate, and severe kidney disease. Because of its mechanism of action, it was observed that Jardiance was not effective in patients with severe renal impairment when the GFR was less than 30 ml/minute.
However, in patients with mild to moderate renal impairment, it significantly improved the glycated hemoglobin by:
- -0.6% in patients with mild renal impairment with a GFR of 60 to 90 ml/minute
- -0.5% when the GFR was 60 to 45 ml/minute, and
- -0.2% when the GFR was 45 to 30 ml/minute.
Like other SGLT2 Inhibitors and especially Dapagliflozin (Farxiga), it significantly decreases urinary microalbumin excretion [Ref].
The manufacturer does not recommend using Jardiance in patients with a GFR of less than 30 ml/minute.
However, in one large cardiovascular outcome trial, a cut-off of 20 ml/minute was used. Patients who had a GFR of 20 ml/minute or more were included in the trial.
Jardiance does not need dosage adjustment if the GFR is more than 45 ml/minute. If the GFR drops to less than 45 ml/minute, a lower dose should be used i.e. 10 mg per day.
It should not be used in patients with a GFR of less than 30 ml/minute and is contraindicated in patients who are on dialysis.
Side effects of Empagliflozin (Jardiance):
Empagliflozin is a relatively safer medicine compared to most other anti-diabetic medicines. However, because of its mechanism of action, it is associated with frequent urogenital infections.
Contraindications to Empagliflozin (Jardiance):
Jardiance (Empagliflozin) is contraindicated in patients who are allergic to the drug. If a person who is allergic to empagliflozin takes it again, he/ she may develop serious symptoms including:
- Shortness of breath, Wheezing, and Cough
- Low blood pressure (Hypotension)
- Fever
- Skin rash (Urticaria) and Itching (Pruritis)
These symptoms may be serious enough and may require intravenous fluids, antihistamines, and/ or adrenaline.
Empagliflozin is also contraindicated in patients who are on dialysis.
Common and uncommon Side effects of Jardiance (empagliflozin):
The two most common side effects of Empagliflozin (Jardiance) are:
- Frequent Urination, and
- Urinary tract infection
Because of the mechanism of action of empagliflozin (Jardiance), urinary frequency is increased in almost all patients.
However, for people with very good blood glucose control, this effect or side effect is not a major problem as empagliflozin is only effective when the blood glucose is high.
On the other hand, for people with high blood glucose, the increased frequency of urination might be one of the factors that they stop taking Empagliflozin (Jardiance).
Increased urinary frequency is also a major problem in people who are using concomitant diuretics and those who have a fluid-overloaded condition.
The other most common side effect of Empagliflozin is urinary tract and genital infection. UTI has been observed in more than 10% of the patients who take Empagliflozin.
UTI is more common in females, those with a previous history of UTI, patients with an immunocompromised condition, and people with uncontrolled diabetes.
Other less common side effects of Jardiance include:
- Acute Renal Failure
- Euglycemic Diabetic Ketoacidosis
- Hypoglycemia
- Dyslipidemias
- Increased risk of fractures
Empagliflozin Brand Names and Price:
Empagliflozin is available worldwide under the brand name Jardiance. Local pharmaceutical companies have started marketing it by various brand names in different countries.
The price of Jardiance in various countries is variable. It costs around USD 500 per 30 tablets in the United States.
Empagliflozin Combination Brand Names:
Empagliflozin is available as:
- Jardiance 10 mg, and
- Jardiance 25 mg.
In combination with other drugs, the following formulations have been marketed:
- Synjardy: It is a combination of Empagliflozin and Metformin
- Glyxambi: It is a combination of Empagliflozin and Linagliptin
- TiJardy XR: It s a combination of Empagliflozin, Linagliptin, and Metformin



